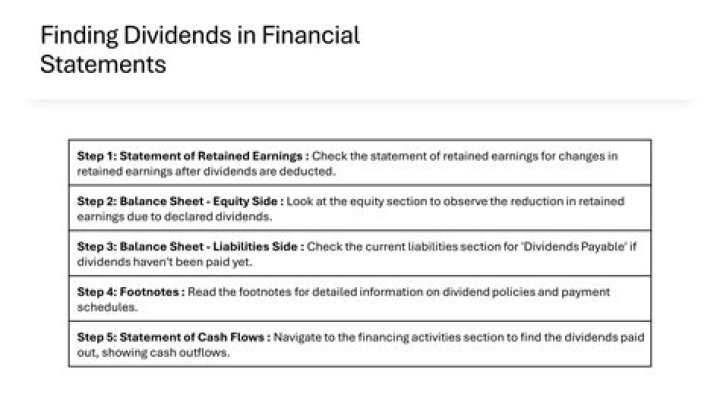
.
Thereof, what type of bonds do noble gases form?
The larger Noble gases have been shown to form some compounds with F and O. An example would be XeF where xenon will form covalent bonds with fluorine. While the Noble gases are relatively non-reactive, they have been forced to form some covalent bonds.
Also Know, how does ionic bonds form? Ionic bond. Ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
Keeping this in consideration, why don t noble gases form ionic compounds?
All noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Because of their high ionization energy and almost zero electron affinity, they were not expected to be reactive.
Why do heavier noble gases form compounds?
Chemists originally believed that the noble gases could not form compounds , because their full valence shell of electrons made them chemically stable and unreactive. But the heavier noble gases have more electron shells than the lighter ones. Since then, many other noble gas compounds have been synthesized.
Related Question AnswersCan noble gases Bond?
2 Answers. Noble gases usually do not form strong bonds between their atoms - it takes fair amount of energy to dimerise them into excimers, but those are short-lived excited molecules. On the other hand there are many stable molecules created by heavier noble gases (mainly xenon) with other elements.Can noble gases form covalent bonds?
The noble gases are the least reactive of all the elements but the heavier ones do form some molecules. Helium and neon never form molecules. They have completely filled electron shells with no have-filled orbitals available for making covalent bonds and they have very high ionization energies so they don't form ions.Are noble gases flammable?
The main properties of the noble gases include: they have low densities. they are inert, so they are not flammable.What makes a gas inert?
An inert gas is a gas that has extremely low reactivity with other substances. The noble gases—helium, argon, neon, xenon, krypton, radon, and element 118 (Uuo)—exist in their elemental form and are found in Group 18 of the periodic table. Inert gases have many practical uses because of their low reactivity.Why halogens are so reactive?
Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements.What makes a noble gas noble?
Noble gas. The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.How many bonds can oxygen form?
2 bondsDo noble gases have electronegativity?
Electronegativity is the ability of an atom to gain an electron. Since the noble gases already have eight electrons in their outer shells, they dont wan't to attract any more. Since electronegativity measures the amount of attraction between an atom and an electron, noble gases do not have electronegativity.Can gases be ionic?
Yes, it may. Because if melting an ionic compound requires extreme temperature, to vaporize it you will need superlative amount of heat energy. So, there you have it – ionic compounds can exist as solid, liquid and gas depending on the conditions they are exposed to.What are the 7 noble gases?
Noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).Are noble gases stable?
Noble gas. The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.What is the difference between ionic and covalent bonds?
The two main types of chemical bonds are ionic and covalent bonds. An ionic bond essentially donates an electron to the other atom participating in the bond, while electrons in a covalent bond are shared equally between the atoms. The only pure covalent bonds occur between identical atoms.What is meant by covalent bond?
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. For many molecules, the sharing of electrons allows each atom to attain the equivalent of a full outer shell, corresponding to a stable electronic configuration.What do noble gases never form?
All noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Their high ionization energy and almost zero electron affinity explain their non-reactivity.Are halogens reactive?
The halogens are all elements that are found in group 17 of the periodic table. The halogens include fluorine, chlorine, bromine, iodine, and astatine. All members of the halogen family have seven valence electrons. Because these atoms are so close to having a full set of eight valence electrons, they're very reactive.Which element is a noble gas?
Noble gas. Written By: Noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).What ions do halogens form?
They get less reactive as we go down the group (so fluorine is the most reactive halogen). In reactions with metals the halogen atoms gain an electron to form ions with a 1− charge. The metal halide formed is an ionic compound. With non-metals, the halogens form covalently bonded molecules.What are some examples of ionic bonds?
Ionic bond examples include:- LiF - Lithium Fluoride.
- LiCl - Lithium Chloride.
- LiBr - Lithium Bromide.
- LiI - Lithium Iodide.
- NaF - Sodium Fluoride.
- NaCl - Sodium Chloride.
- NaBr - Sodium Bromide.
- NaI - Sodium Iodide.