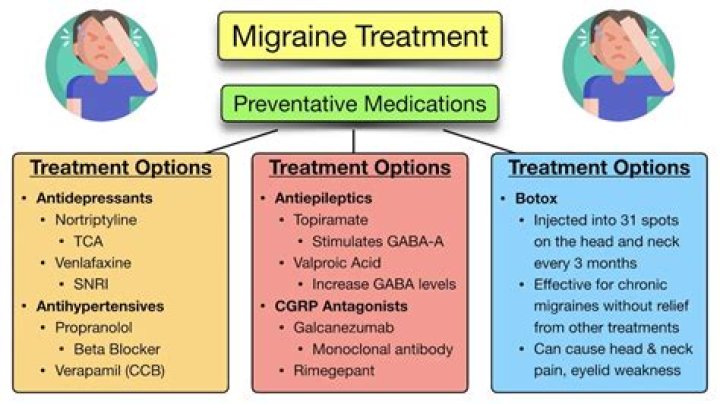
Calcium carbonate is strongly heated until it undergoes thermal decomposition to form calcium oxide and carbon dioxide. The calcium oxide (unslaked lime) is dissolved in water to form calcium hydroxide (limewater). Bubbling carbon dioxide through this forms a milky suspension of calcium carbonate..
Beside this, what is the difference between calcium oxide and calcium carbonate?
Calcium oxide (CaO) is a solid which when wet changes into calcium hydroxide (Ca(OH)2), a strong base. It actually is formed by heating chalk/limestone, which is mostly CaCO3. Calcium oxide (CaO) is a solid which when wet changes into calcium hydroxide (Ca(OH)2), a strong base.
how do you use calcium carbonate? Calcium carbonate is a dietary supplement used when the amount of calcium taken in the diet is not enough. Calcium is needed by the body for healthy bones, muscles, nervous system, and heart. Calcium carbonate also is used as an antacid to relieve heartburn, acid indigestion, and upset stomach.
Consequently, how do you make calcium oxide?
Calcium oxide can be produced by thermal decomposition of materials like limestone or seashells that contain calcium carbonate (CaCO3; mineral calcite) in a lime kiln. The process that is used to prepare burnt lime is known as calcination.
What is the decomposition of calcium carbonate?
CaCO3 (s) ---> CaO (s) + CO2 (g) All metal carbonates decompose to give the metal oxide and carbon dioxide. As calcium ions has a 2+ charge, the formula of calcium carbonate will Be CaCO3 (as carbonate is a 2- ion).
Related Question Answers
Where do you get calcium carbonate?
Calcium carbonate is often found in sea shells and bones of sea creatures, and when these settle on the ocean floor for millions of years it forms limestone. Limestone can also come from the combination of soluble calcium (Ca) and carbon dioxide (CO2) from water in lakes and oceans.What is the common name for calcium carbonate?
Common Name For Calcium Carbonate. Calcium carbonate is a chemical compound with the chemical formula CaCO3. It is a common substance found in rock in all parts of the world, and is the main component of shells of marine organisms, snails, pearls, and eggshells.Is chalk a calcium carbonate?
Composition: Chalk is a form of calcium carbonate, having the same chemical composition as ground calcium carbonate, limestone, marble, and precipitated calcium carbonate (PCC). In fact, all of the calcium carbonates listed in the previous sentence have the same crystal form, calcite.Why is calcium carbonate better than calcium hydroxide?
However, over time, soils tend to become more acidic than this. Calcium carbonate and oxide and hydroxide all react with acids. Most agricultural lime is calcium carbonate because it is cheaper and safer to handle. Because it isn't water soluble it will be slower acting than calcium hydroxide.Why is calcium carbonate important?
Calcium carbonate decomposes to form carbon dioxide and lime, an important material in making steel, glass, and paper. Because of its antacid properties, calcium carbonate is used in industrial settings to neutralize acidic conditions in both soil and water.What are the different forms of calcium carbonate?
Calcium carbonate. Three types of calcium carbonate-containing rock are excavated and used by industry. They are limestone, chalk and dolomite. Limestone and chalk are both forms of calcium carbonate and dolomite is a mixture of calcium and magnesium carbonates.Is Lime a calcium carbonate?
a. Lime is produced by heating limestone in a kiln to change the mineral into calcium oxide, and calcium carbonate (aka: pulverized limestone or fine grind) is produced by pulverizing high-grade calcium minerals, in Pete Lien & Sons' case limestone, into a ground granular or power sized finished product.What is calcium oxide used for?
Calcium oxide is widely used in industry, e.g., in making porcelain and glass; in purifying sugar; in preparing bleaching powder , calcium carbide, and calcium cyanamide; in water softeners; and in mortars and cements. In agriculture it is used for treating acidic soils ( liming ).Is calcium oxide an acid or base?
Calcium oxide (CaO) , commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature.What is the Colour of calcium carbonate?
white
What is the pH of calcium oxide?
It is soluble in acids, glycerol and a sugar solution. If dissolved in water (Ca(OH)2) exhibits a pH of 12.5. CaO has the cubic “halite” structure.Is calcium oxide safe?
It is considered a health hazard, and contact with moist skin or eyes can cause severe irritation. Calcium oxide is highly reactive with water, forming calcium hydroxide, which is also corrosive. Safe storage practices of both calcium oxide and calcium hydroxide are crucial.What type of chemical is calcium chloride?
inorganic compound
Why is it called quicklime?
Calcium oxide, CaO, also known as lime or more specifically quicklime, is a white or grayish white solid produced in large quantities by roasting calcium carbonate so as to drive off carbon dioxide. At room temperature, CaO will spontaneously absorb carbon dioxide from the atmosphere,…Is calcium oxide natural?
Calcium Oxide. Lime does not occur naturally since it reacts so readily with water (to form hydrated lime) and carbon dioxide (to form limestone). It is produced in very large quantities synthetically, however, by the heating of limestone.What is the common name for calcium hydroxide?
slaked lime
What happens when calcium oxide reacts with water?
When water is added to calcium oxide (quicklime), it will be converted to calcium hydroxide (slaked lime) with the liberation of enormous amount of heat. It is an exothermic reaction. Calcium hydroxide is an alkali.What's wrong with calcium carbonate?
Calcium carbonate may cause acid rebound: the stomach overcompensates for the high dose of calcium carbonate, which is alkaline, by churning out more acid. For that reason, people with a history of stomach ulcers are advised that they may not tolerate it and may have to switch to calcium citrate. Constipation.Is baking soda a calcium carbonate?
It seems that baking soda can be used for everything. It comes out of the ground in the form of minerals nahcolite and trona, which are refined into soda ash (a.k.a. calcium carbonate), then turned into baking soda (a.k.a. sodium bicarbonate), among other things.