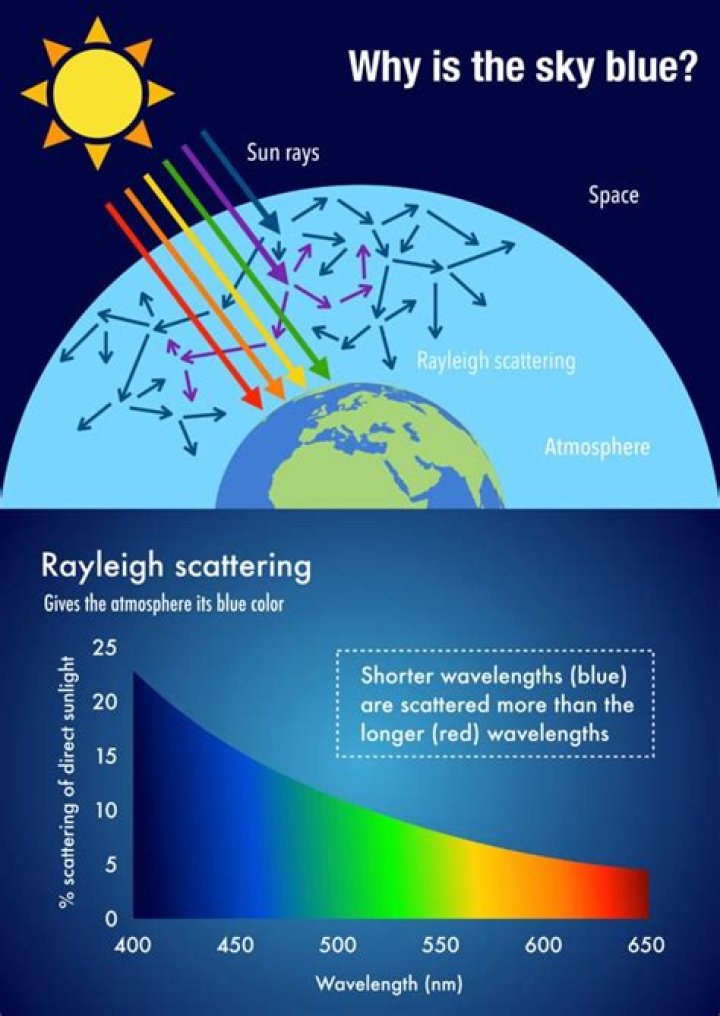
Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. When one of these solids dissolves in water, the ions that form the solid are released into solution, where they become associated with the polar solvent molecules..
Also, why is sugar highly soluble in water?
A Water-soluble sugar The reason glucose dissolves readily in water is because it has lots of polar hydroxyl groups which can hydrogen-bond with water molecules. Hydrogen bonds are very important intermolecular forces which determine the shape of molecules like DNA, proteins and cellulose.
Additionally, what is it called when sugar dissolves in water? When you stir a spoonful of sugar into a glass of water, you are forming a solution. This type of liquid solution is composed of a solid solute, which is the sugar, and a liquid solvent, which is the water. As the sugar molecules spread evenly throughout the water, the sugar dissolves.
Accordingly, why is sugar not infinitely soluble in water?
Solute-solute interactions in sugar are strong enough to make sugar a solid at room temperature. Water molecules must work hard to overcome these interactions and pull sugar molecules away from one another, which diminished the solubility.
Why is sugar more soluble than salt?
Sugar melts easily because the bonds that hold those molecules together in the solid (crystalline state) are much weaker. The initially stronger ionic bonds in salt and the more numerous water-sugar bonds that form after dissolution gives sugar the solubility edge. Thus the greater solubility of sugar over salt.
Related Question Answers
Why are monosaccharides water soluble?
Monosaccharides are quite soluble in water because of the numerous OH groups that readily engage in hydrogen bonding with water.What makes something water soluble?
When polar compounds or ions are added to water, they break up into smaller components, or dissolve, to become part of the solution. The water's partial charges attract different parts of the compound, making them soluble in water.Is glucose water soluble?
Water Acetic acidDoes ethanol dissolve in water?
Ethanol has a polar –OH group, which hydrogen bonds to water; which makes ethanol soluble. Ethane which consists of Ions of carbon and hydrogen only has no polar group and is not water-soluble.Why is starch insoluble in water?
Starches are insoluble because they exist as granules. Amylopectin is not less soluble because of its branched chains of glucose, it is actually more soluble with amylose causing stabilization & locking the crystalline structure to make the granule even stronger.Why is nacl soluble in water?
Salt (sodium chloride) is made from positive sodium ions bonded to negative chloride ions. Water can dissolve salt because the positive part of water molecules attracts the negative chloride ions and the negative part of water molecules attracts the positive sodium ions.Why Some salts are insoluble in water?
Insoluble salts are ionic compounds that are insoluble in water: the salt continues to exist as a solid rather than dissolving in the liquid. When a salt such as sodium chloride (table salt) dissolves in water, its ionic lattice is pulled apart so that the individual sodium and chloride ions go into solution.Is starch water soluble?
Starch becomes soluble in water when heated. The granules swell and burst, the semi-crystalline structure is lost and the smaller amylose molecules start leaching out of the granule, forming a network that holds water and increasing the mixture's viscosity. This process is called starch gelatinization.What does infinitely soluble mean?
"Solubility" is often expressed in "grams of solute per 100 grams of solvent". When "substances" are "infinitely soluble" with each other it means that they will dissolve any amount of each other until they reach the "saturation point" with had trouble for example, water and ethanol are "infinitely soluble".Why sugar is soluble in water?
It also takes energy to break the hydrogen bonds in water that must be disrupted to insert one of these sucrose molecules into solution. Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules.Why is oil not soluble in water?
Many substances do not dissolve in water and that is because they are non-polar and do not interact well with water molecules. A common example is oil and water. Oil contains molecules that are non-polar, thus they do not dissolve in water.What determines if a substance will dissolve?
compositions of a solvent/solute determine whether or not a substance will dissolve. Stirring (agitation) temperature and surface area of dissolving particles determine how fast the substance will dissolve. maximum amount of solute for a given quantity of solvent at a constant temperature and pressure.Why do small particles dissolve faster?
Dissolving is a surface phenomenon since it depends on solvent molecules colliding with the outer surface of the solute. A given quantity of solute dissolves faster when it is ground into small particles than if it is i n the form of a large chunk because more surface area is exposed.What conditions cause solids to dissolve faster?
By stirring a gas in a liquid, its solubility (speeds up, slows down) the dissolving process. A solid dissolves faster in a liquid if the temperature of the liquid is (increased, decreased). The (larger, smaller) the surface area of a solid, the faster it will dissolve.What makes a substance dissolve faster in a solvent?
The particle size of solute particles affects the rate. Stirring a solute into a solvent speeds up the rate of dissolving. This is because it helps distribute the particles throughout the solvent. For example, when you add sugar to iced tea and then stir the tea, the sugar will dissolve faster.What describes liquids that are soluble in each other?
When two liquids can be readily combined in any proportions, they are said to be miscible. An example would be alcohol and water. Either of the two can totally dissolve each other in any proportion. Two liquids are defined as immiscible if they will not form a solution, such as oil and water.How will you find out whether a solute dissolves in water?
The best way to tell if something will dissolve is to look at the polarities of the solvent and the solute. If the polarities of the solvent and solute match (both are polar or both are nonpolar), then the solute will probably dissolve.How do you dissolve sugar in water?
The sugar cubes soften when placed in the water and begin to dissolve. The sugar cube in the hot water dissolves the fastest. Sugar will dissolve faster when you stir the solution quickly because the act of stirring increases kinetic energy which increases the temperature.What happens to sugar when mixed with water?
the molecules have broken down into atoms and dispersed in the water. The sugar molecules cannot go away but they can disperse in the water. They will still be sugar molecules just not attached to any other molecules of sugar. the water and the sugar particles will be mix together and form a new substance.